Product information
BCG Apogepha® 100 mg
Active ingredient: Bacillus Calmette-Guérin (BCG)
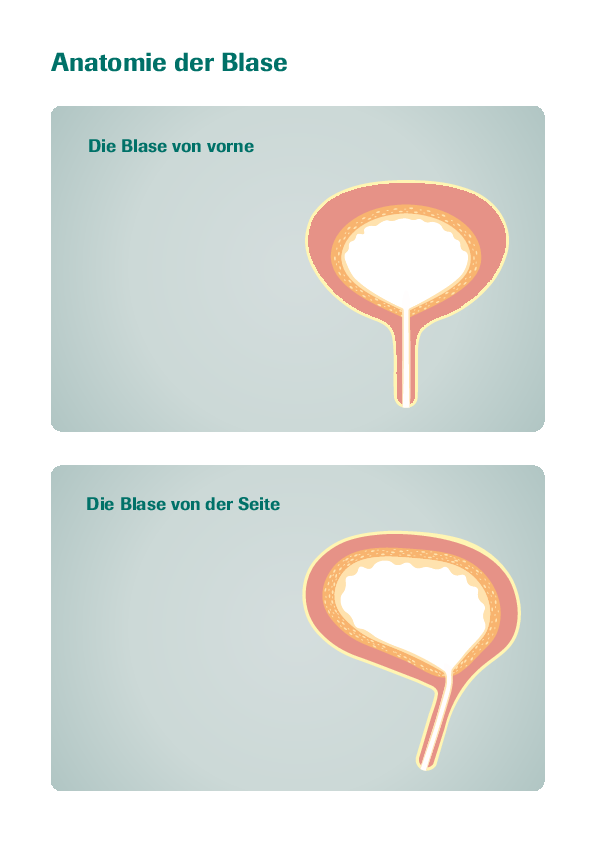
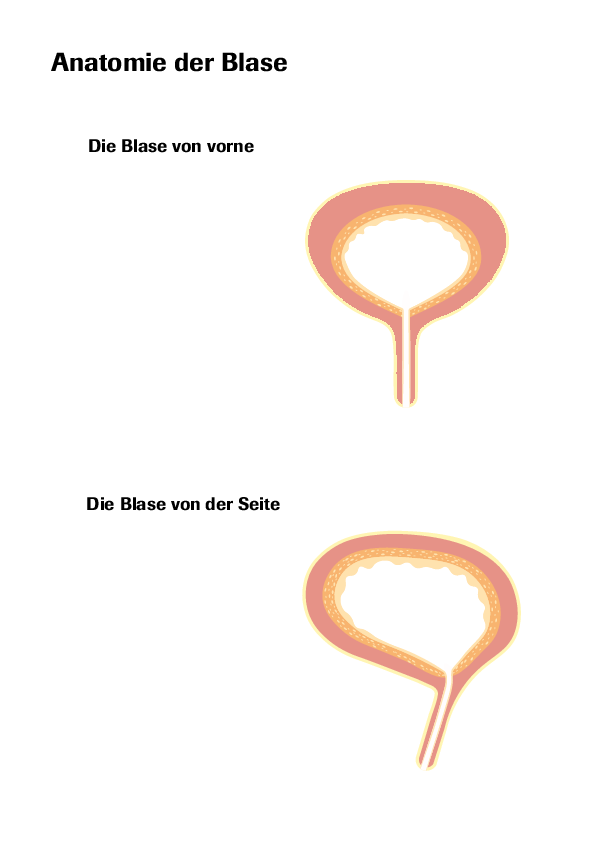
Indication: BCG Apogepha® 100 mg is intended for intravesical use (inside the bladder) to treat superficial bladder cancer following transurethral resection (removal of tissue through the urethra).
Dosage, type and duration of use: BCG Apogepha® 100 mg should only be used by doctors who have experience with this treatment. There is one treatment regimen for BCG, including 6 weeks of induction therapy and 1–3 years of maintenance therapy. The exact duration may vary and must be assessed on a case-by-case basis.
Specialist group information
Click here to go to the specialist group login. You can see more detailed information after logging in.
Pack sizes
Pack sizes of BCG Apogepha® 100 mg
BCG Apogepha® 100 mg is available in N1 and N2.

| PZN (pharmaceutical identification no.) | Pack size | Standard size |
|---|---|---|
| 20023138 | 1 vial + 1 bladder instillation system | N1 |
| 20023121 | 3 vials + 3 bladder instillation systems | N2 |
Free material for download
Free material for download
With just one click, you can download the usage and technical information, as well as other service material: